|
At the 75 mg QD dose level (n =4), drug-related grade 2 adverse events were reported which led to dose reduction to 65 mg QD (n=3) or treatment discontinuation (n=1). The 65 mg cohort was expanded to a total of 10 patients, and no other DLTs were observed. One subject treated at 65 mg experienced a DLT of grade 3 rash. Drug-related grade 3 or 4 TEAEs included neutrophil count decreased (n=3), as well as febrile neutropenia, cellulitis, platelet count decreased, lipase increased, and rash (one each). The majority of the drug-related TEAEs were grade 1 or 2. The most common drug-related TEAEs that occurred in > 2 patients were nausea (n=4), diarrhea (n=4), fatigue (n=3), neutrophil count decreased (n=3), dysgeusia (n=3) and rash (n=3). Enrolled patients received ARQ 531 orally once daily, continuously, in 28- day cycles at doses of 5, 10, 15, 20, 30, 45, 65 and 75 mg QD. 
BTK-C481S mutation was documented in 22/26 (85%) CLL patients. Baseline demographics were: median age 65.5 (range 47-82) years, male/female 36/4 and median number of prior lines of therapy 4 (range 2-12). Results: As of July 19, 2019, a total of 40 patients have been treated: CLL/SLL (n=26), Richter's transformation (n=6), DLBCL (n=3), FL (n=4), MCL (n=1). Tumor responses were evaluated per disease specific guidelines. Treatment emergent adverse events (TEAEs) were assessed per NCI CTCAE v.4.03. Dose escalation was performed according to a 3+3 study design. Prior therapy for CLL must have included an irreversible BTK inhibitor. Eligible patients had relapsed/refractory CLL/SLL, B-cell NHL or Waldenstrom's macroglobulinemia, had received at least 2 prior lines of systemic therapy and had good organ function including creatinine clearance ≥ 60 mL/min as estimated by the Cockcroft-Gault equation or by 24-hour urine collection, absolute neutrophil count ≥ 1000/µL, platelet count ≥ 50,000/µL and hemoglobin ≥ 8.0 g/dL. The secondary objectives were to assess the pharmacokinetic (PK) profile, pharmacodynamic (PD) activity, and preliminary evidence of anti-tumor activity. 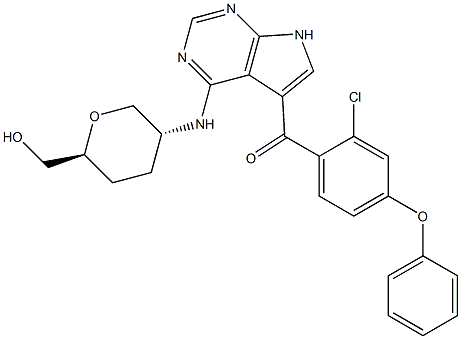 
Methods: The primary objectives of the clinical study were to assess the safety and tolerability of ARQ 531, and to determine the recommended Phase 2 dose (RP2D) and schedule. ARQ 531 suppresses oncogenic BCR signaling in CLL cells resistant to ibrutinib and has demonstrated antitumor activity superior to ibrutinib in CLL, Richter's transformation, and DLBCL mouse models. ARQ 531 is a potent, reversible inhibitor of both wild type and ibrutinib-resistant C481S-mutant BTK. Introduction: Resistance to covalent BTK inhibitors such as ibrutinib and acalabrutinib is a common mechanism of resistance that portends a poor long-term clinical outcome.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
